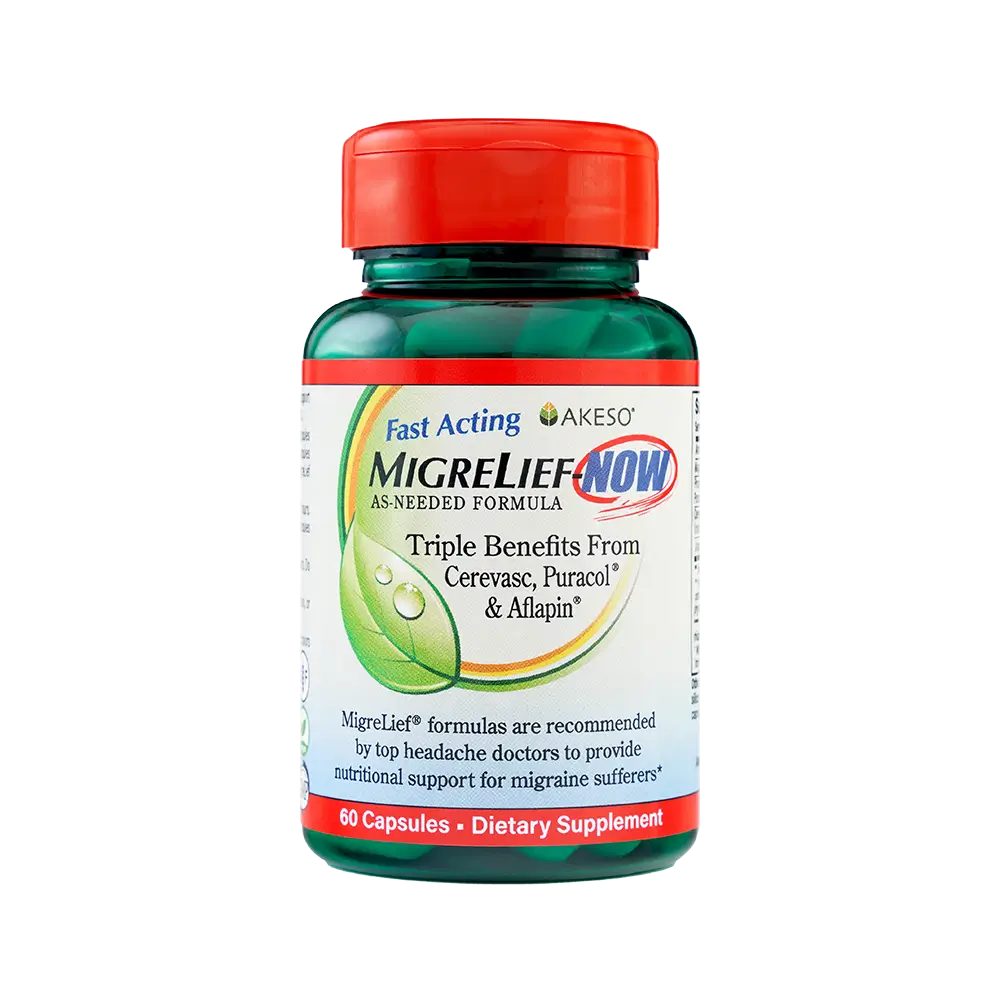
MigreLief-NOW
A fast-acting, as-needed dietary supplement to provide neurological comfort and support for migraine sufferers.*

Migraine Support You Deserve
Frequently Asked Questions
MigreLief-NOW is a fast-acting, as-needed migraine supplement for use at first warning signs. It may be combined with any daily MigreLief formula or used alone for additional nutritional support as needed.
MigreLief-NOW contains Magnesium (180 mg), Puracol® Feverfew (50 mg), Cerevasc™ Ginger (500 mg), and Aflapin® Boswellia Serrata (100 mg) per 2-capsule serving. The ingredients are vegetarian, free from drugs, and caffeine.
Take 2–4 capsules with food at the first sign of discomfort. You may repeat the dose in 2 hours if needed, but do not exceed 8 capsules in 24 hours for adults if taking a daily MigreLief supplement as well. For children, dosing varies by age (see product label for details).
MigreLief-NOW is appropriate for adults and children over age 2, but dosages vary by age. See back of label.
It is formulated as-needed nutritional support during difficult times.
No harmful side effects are typical, but discontinue use if allergic to any ingredients.
Yes. MigreLief-NOW is safe for occasional or even daily use, but always follow dosing instructions.
You Have a Choice of Four MigreLief Formulas
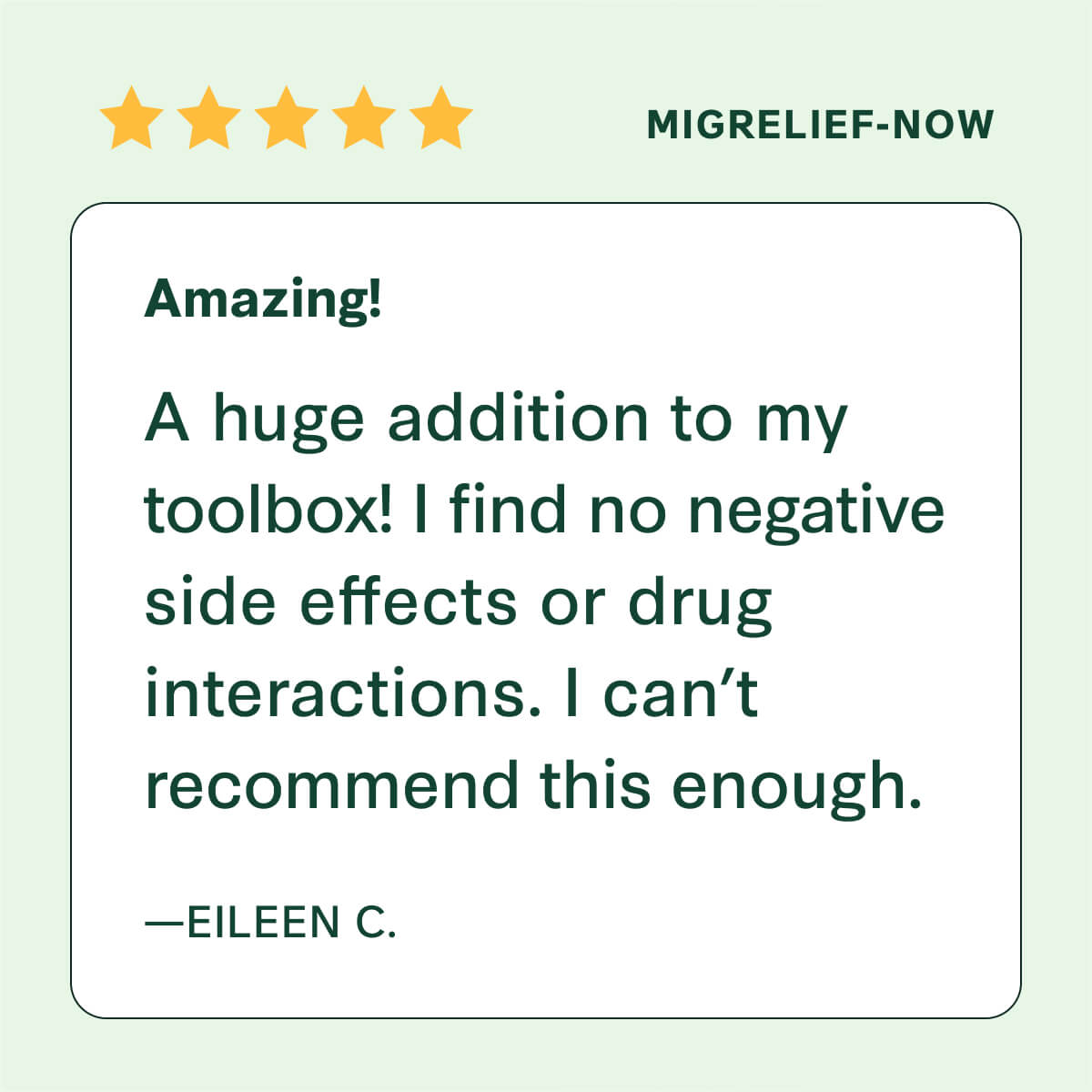
Trusted By Thousands
Reviews Disclaimer
The product reviews below are not a substitute for professional medical advice, diagnosis, or treatment. The opinions below reflect the views and opinions expressed by our customers and not those of MigreLief, and are provided for informational purposes only. We do not verify or endorse any claims made in these reviews.